- How Many Electrons Are In Oxygen
- Number Of Electrons In Sodium Cation
- Number Of Electrons In Sodium 24
In an atom, the electrons spin around the center, also called the nucleus. The electrons like to be in separate shells/orbitals. Shell number one can only hold 2 electrons, shell two can hold 8, and for the first eighteen elements shell three can hold a maximum of eight electrons. Also, how many electrons does sodium atomic number 11 need to lose to be full? If sodium (group 1) loses 1 electron, it gets a full outer shell. Because the sodium ion then has 11 protons but only 10 electrons, it gains a posi)ve charge of +1. Calcium, the next atom along, needs to lose 2 electrons to get a full outer shell, giving it a charge. Since, each Na atom contributes one electron, the total number of electrons in 1 cm 3 (n) = 2.544 x 10 22 electrons. Given that the particles's mean separation d = 1/n 1/3 (it should be noted that it is an approximation) Plugging the value of n gives d = 3.4 x 10-8 cm or 0.34 nm. A neutral atom has the same number of protons and electrons (charges cancel each other out). An ion has an unequal number of protons and electrons. If the charge is positive, there are more protons than electrons. If the charge is negative, electrons are in excess. Deleting evernote account. You can find the number of neutrons if you know the isotope of the atom.
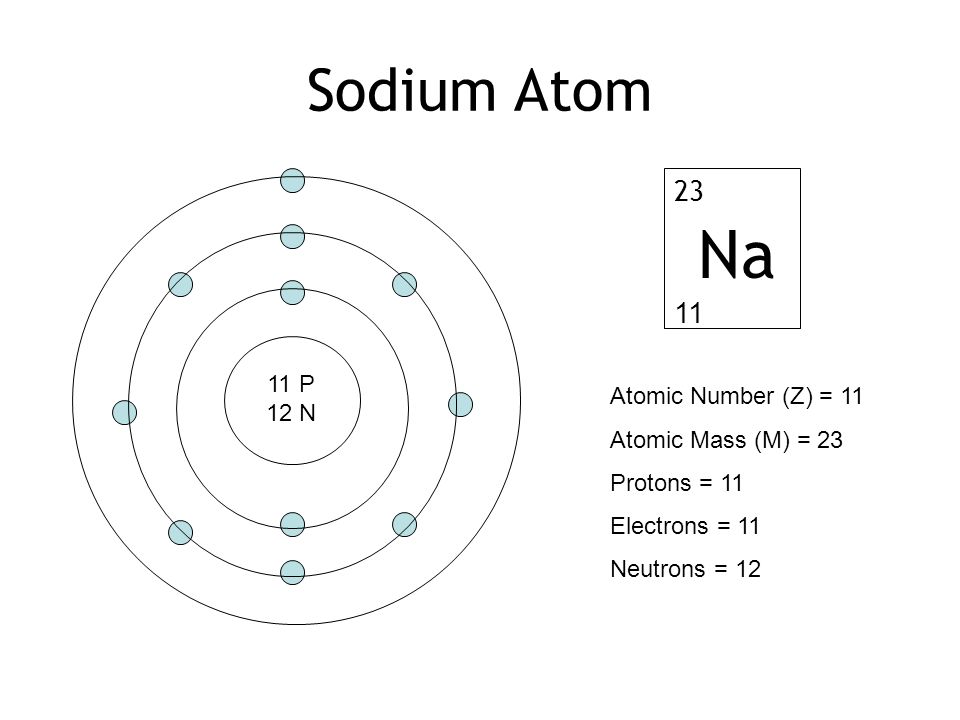
So for your question, the Periodic Table tells us that sodium has an Atomic Number of 11, so there are 11 protons and 11 electrons. The Periodic Table tells us that sodium has an Atomic Mass of ≈23. So there are 23 - 11 = 12 neutrons.

Click to see full answer
Also know, how many protons neutrons and electrons does sodium have?
The element sodium has 12 neutrons, 11 electrons and 11 protons. The number of electrons and protons come from the element's atomic number, which is same 11. The number of neutrons can be found by subtraction of the atomic number from sodium's atomic mass of twenty three.
Secondly, how many protons electrons and neutrons are in a given element? The atomic number (number at the top) is the amount of protons and the amount of electrons. So if an element has an atomic number of 5, you know that it has 5 protons and 5 electrons. The atomic mass (number at the bottom) is the amount of protons and neutrons added together.
How Many Electrons Are In Oxygen
Also to know is, how many neutrons are in an atom of sodium?
11
Number Of Electrons In Sodium Cation
How do you find the number of neutrons in sodium?
Number Of Electrons In Sodium 24
The mass number of an element tells us the number of protons AND neutrons in an atom (the two particles that have a measurable mass). Sodium has a mass number of 23amu. Since sodium has 11 protons, the number of neutrons must be 23 – 11 = 12 neutrons.
